
Back

|
The Serpin Database Back |
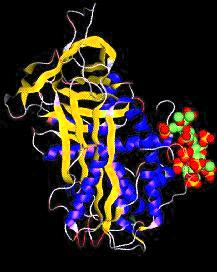 |
| Antithrombin with bound pentasaccharide.
Click on the picture for VRML tour of serpin structure |
Notably, the serpins are the principle protease inhibitors in human plasma :
Altogether the inhibitory serpins make up some 10% in molar terms, of the proteins in human plasma. Also present, in plasma, though in smaller concentrations, are other serpins that have lost their inhibitory activity but have taken on other functions vital to life; examples are the vasopressor peptide source angiotensinogen, and the thyroxine and corticosteroid binding globulins, TBG and CBG.
The reason for the evolutionary success of the serpins is their possession, uniquely amongst the many families of serine protease inhibitors, of a mobile reactive site loop. It is the ability of this loop to profoundly change its conformation that enables the serpins to bind to their target proteases as a virtually irreversible complex.