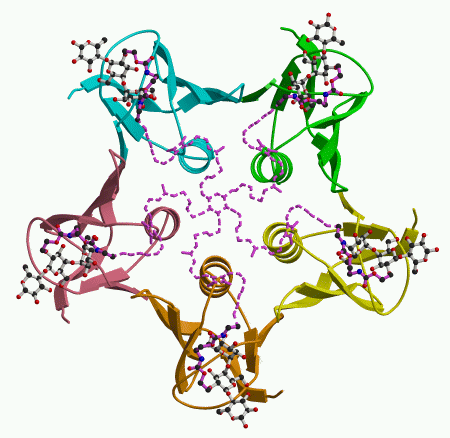
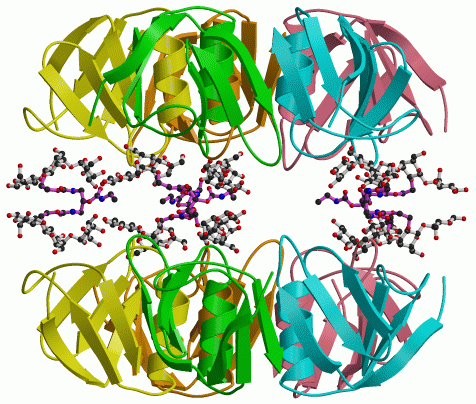
To get feedback on the design of the starfish molecule, we looked at how it binds to the Shiga-like toxin B pentamer. This showed us that the design had worked in part, but there was an interesting surprise. The arms of the starfish molecule do indeed reach out to the five subunits of the B pentamer, as shown in the figure on the left. (Atoms drawn with dashed lines could not actually be seen, but their approximate locations could be inferred.) However, we found that the Pk-trisaccharide components bind only at site 2. Instead of binding to site 1 on the same toxin molecule, the trisaccharide connected by the bridge binds to site 2 on a second pentamer, as shown in the figure on the right. So one starfish molecule glues two toxins together, taking them both out of action.
 |
 |
Remember that site 2 is the site most similar to the one seen in cholera toxin. This suggests that the design of the starfish molecule could be adapted to inhibit the action of cholera toxin. More generally, the success of the starfish molecule demonstrates that multivalent carbohydrate inhibitors can be designed successfully.
The next step will be to see whether the starfish molecule, or something like it, can be used to treat patients infected with E. coli producing Shiga-like toxins, to prevent HUS. It is a promising lead, but much remains to be done.
|
Back The starfish inhibitor: molecular Velcro |
Up Back to top |
Next Publications |